Negative temperature???
But how are negative temperatures hotter than positive ones?
First of all, when I say negative temperatures, I don’t mean like -1°C, or -200°C, because those are actually still positive, because they have a positive amount of heat, it’s not like 0°C has nothing and -1°C has some cold and 1°C has some heat, they all have heat, so the true 0° of temperature would have no heat at all, and that is represented by a different scale of temperature, where 0°K (Kelvin) is actually 0 temperature, in the way that it has no heat energy at all.
Secondly, before we get into negative temperature, I must tell you what temperature is actually measuring, and in what way it’s measuring it.
The thing temperature is measuring in a substance is the amount of the substance’s atoms that are in their highest energy state as opposed to its atoms in their lowest energy state, so the coldest (0K) is where all the atoms are in their lowest energy state, and the ‘hottest’ (Infinity Kelvin) is where, you didn’t guess it, there are as many atoms in the highest and the lowest energy states.
So you’d think that where the ratio is 1:1 highest:lowest (where the lowest is always represented by 1 in the ratio), then that would be 1°K, NO! It’s actually Infinity°K!
And then, you might think, what if MORE atoms are in the highest energy state than lowest energy state, then what would the temperature be? Negative! Like what how does that make sense? And what’s more, if you have more and more atoms in the highest energy state, approaching a configuration where all of them are in the highest energy state, the temperature in °K approaches 0 from the negative side.
Okay, so how does any of this make sense.
Well, if you graph out 1/x, where the height of each point in the graph is just it’s x position (how far right it is, left would be the opposite of right so would be a negative x position) , but 1 divided by it, so the point in the graph that is 1 unit to the right of the middle, for example, would actually also be 1 unit high, because 1/1 = 1. At 1/2 a unit it would be 2 high, at 1/10000 a unit it would be 10000 high, and so on.
But notice this.
When you have some amount of temperature, like let’s start at room temperature for example, 275.15K, not too big or small of a number, you can heat it and heat it, but never to infinite temperature, where half the atoms are in their highest energy state, and you can cool it down closer and closer to 0K, where there is no energy at all, but you can’t actually GET there either, so it’s infinitely hard to get to those two temperatures, the two extremes.
But look, if you look at the graph of 1/x, and start at like where the HEIGHT of the graph is 1 for example, and follow along the line of the GRAPH, NOT just straight left or right or any directione, and you keep going left for example, towards where the height is infinity, (ALONG THE GRAPH THOUGH, REMEMBER) you can get closer and close, but you can’t actually GET to infinity, because you would have to go forever and ever up that ever-steeper graph line, because the height is infinite, and similarly, if you go the other way, towards zero, then you can get closer and closer, but you can never get there still, because it’s also infinitely far away along the ever-flattening graph, because you would have to go infinitely far right to get a height of 0 on this graph.
And for the negative temperatures, they’re actually hotter, because they have even more atoms in their highest energy state, and the reason we call the hottest temperature -0 degrees kelvin is because as you get closer and closer to the state that all atoms are in their highest energy state (which, by the way, is impossible to get to exactly and perfectly), the temperature (and actually also the graph of 1/x), gets closer and closer to 0, but from the negative side, like gets smaller and smaller negative numbers.
So now, I feel you can see why these temperatures are represented like that.
But can we just appreciate and see, that in this system, when we approach the state of half the atoms in the highest energy state from a state with less, so that it increases to the state with half the atoms in the highest energy state, the temperature goes higher and higher infinitely, but if you get to that state from a state with negative temperature, where there were MORE than half of the atoms in the highest energy state, then it gets bigger and bigger negative temperatures. Just like the graph 1/x. And the lines diverging in gradient but approaching the same line that I talked about last post. It all points to the idea going to ‘positive infinity’ and ‘negative infinity’ are ACTUALLY BOTH GOING TO THE SAME NUMBER! Because loads of places where it actually appears, in a comprehensible context, it really seems like it’s the exact same thing.
So yeah, that’s why the temperature system is like that and that’s how it makes sense

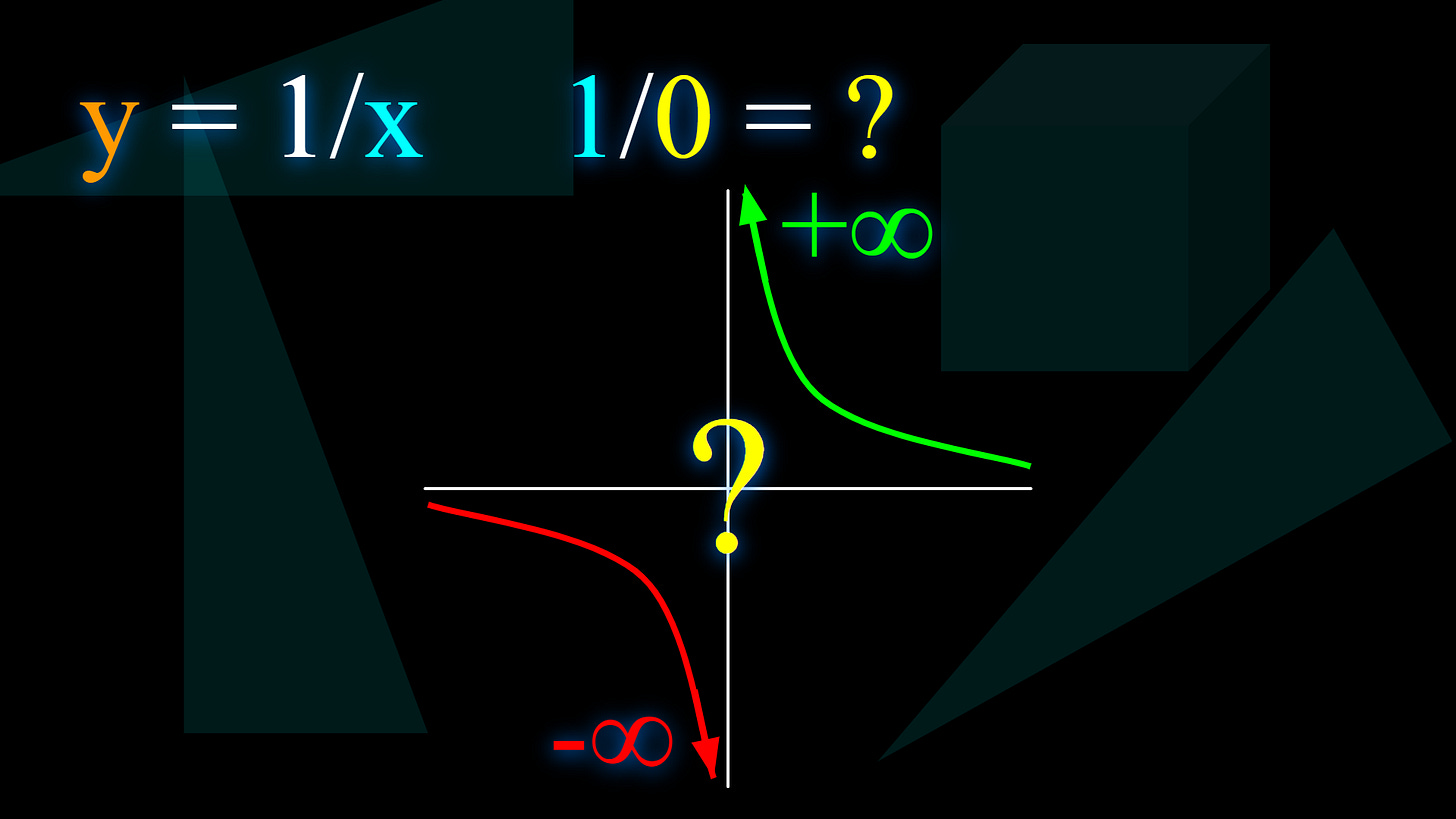
Only a truly Brilliant mind can analyse it thus!
😳😳😳🤯🤯🤯